|
Number of four bonds around carbon atoms, if carbon form a stable compound. Chlorine's minimum valence is 1 and carbons minimum (only) valence is 4.Now, we are going to consider the minimum valence of chlorine and carbon atoms to form stable compounds.Now, there is a confusion between how to select the center atom.In that case, we can suggest that carbon has the potential to be the center atom. Therefore, carbon is more electropositive than chlorine. Most electropositive element - Carbon's electronegativity is 2.5 and chlorine's electronegativity is 3.0Īccording to the Pauling's electronegativity scale.We can suggest that chlorine has the potential to be the center atom. Considering greater valence - Chlorine's highest valence is 7 and carbon's highest valence is 4.To be the center atom, ability of having greater valance and being most electropositive element in theīecause, CCl 4 is a simple molecule, it is not very difficult to select the center atom. For,ĬCl 4, total pairs of electrons are 16 (32/2) in their valence shells.Ĭenter atom selection and draw basic skeletal of CCl 4 Total electron pairs are determined by dividing the number total valence electrons by two. Pairs = σ bonds + π bonds + lone pairs at valence shells valence electrons given by chlorine atoms = 7 * 4 = 28.valence electrons given by carbon atom = 4 * 1 = 4.Now we know how many electrons are included in There are only two elements in CCl 4 molecule carbon and chlorine.Įlement in the periodic table and contains 7 electrons in its last shell.Ĭarbon atom has 4 electrons in its last shellīecause it is a IV group element. Total number of electrons of the valance shells of CCl 4 Check the stability and minimize charges on atoms by converting lone pairs to bonds to obtain best.Mark charges on atoms if there are charges.Find center atom and draw basic skeletal structure.Determine total electrons pairs as lone pairs and bonds.Find total number of electrons of the valance shells of carbon atom and chlorine atoms. 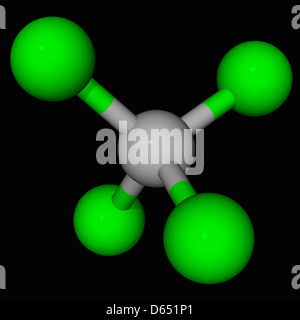
Each step of drawing is explained in detail in this tutorial. There are several steps to complete the lewis structure of CCl 4. 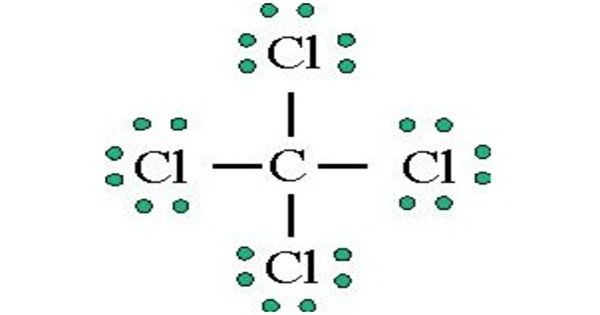 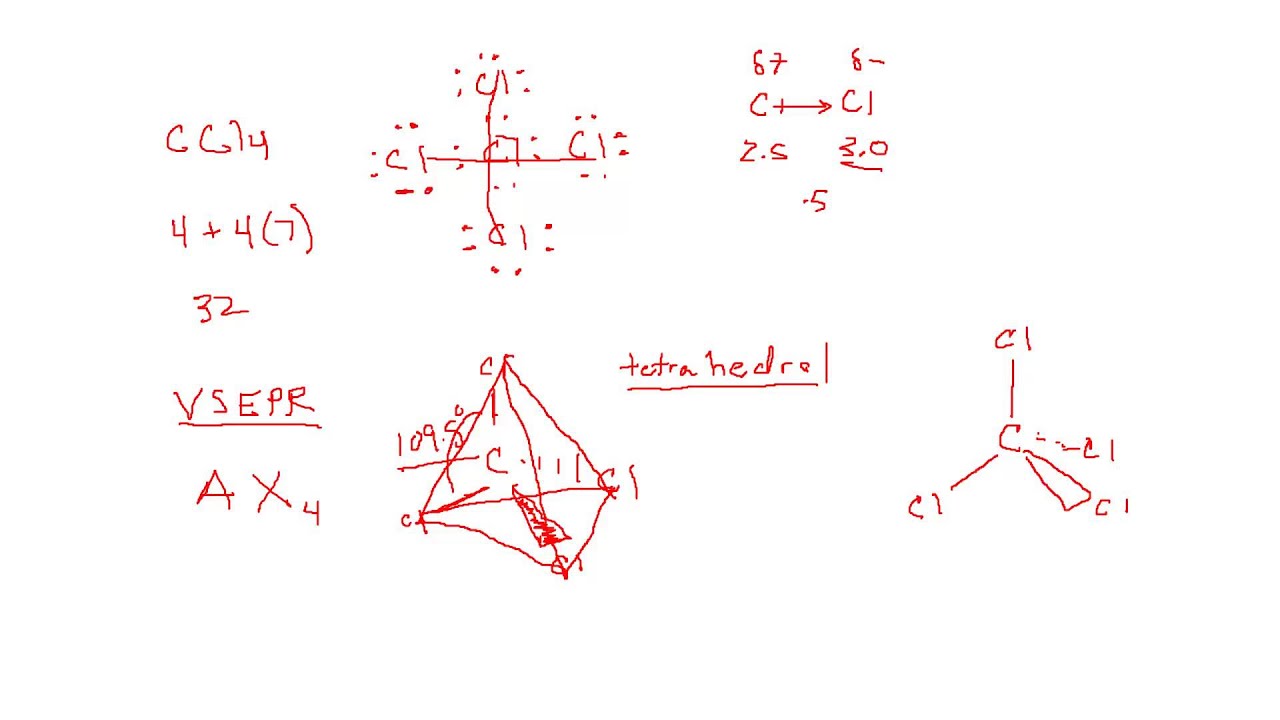
Steps of drawing lewis structure of CCl 4 Carbon tetrachloride is a clear colorless liquid at room temperature and a noncombustible compound.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
